
This page consists of Science CBSE sample papers for class 9. A downloadable copy of this page is also available at the bottom of the page. So all the best for your exams. If you like the content do not forget to like share or comment at the section given below. you will find extra resource links (science study material) at the end of the page.
Section A
Question 1
A horse of mass 210 Kg and a dog of mass 25 Kg are running at the same speed. Which of the two possesses more Kinetic energy? How? (2 Marks)
Question 2
Define ozone hole? (2 Marks)
Question 3
Seema tried to push a heavy rock of 100 Kg for 200 s but could not move it. Find the work done by Seema at the end of 200 s. ( 2 Marks)
Question 4
Which group of plants has nitrogen-fixing bacteria in the root nodules? (2 Marks)
Question 5 (3 Marks)
(i)Name the fundamental particle not present in the nucleus of hydrogen atom.
(ii)Give one important application of isotopes of cobalt and uranium.
Question 6 ( 3 Marks)
(i)What is notochord?
(ii)Mention its functions.
Question 7 (3 Marks)
(i)Why does a block of wood held under water rise to the surface when released?
(ii)An object of weight 200 N is floating in the liquid. What is the magnitude of buoyant force acting on it?
Question 8 (3 Marks)
(i)Three persons A, B and C are made to hear a sound travelling through different media as given below. Who will hear the sound first and why?
| Person | Medium |
| A | Iron rod |
| B | Air |
| C | water |
Question 9 (3 Marks)
(i)What is soil erosion?
(ii)List two activities which cause soil erosion.
(iii) Sound of explosion taking place on other planets is not heard by a person on earth. Give reason.
Question 10 (4 Marks)
(i)Write chemical formulae of the following compound
(a)Aluminium nitride
(b)Ammonium phosphate
(ii)Name the metal which shows variable Valency. Write the formulae of two of its chlorides.
Question 11 (3 Marks)
Thallophyta, bryophyte and pteridophyte are classified as cryptogammae whereas gymnosperms are classified a phanerogammae, why?
Question 12 ( 3 Marks)
Define relative density of a substance. Relative density of silver is 10.8. The density of water is 1000 kg/m3. What is the density of silver in SI units.
Question 13 (3 Marks)
(i)Define air pollution.
(ii) Write two methods to prevent it.
Question 14 ( 6 Marks)
Write the most striking features of the following phya/class?
(i)Arthropoda
(ii)Amphibia
(iii)Porifera
Question 15 (3 Marks)
(i)Calculate the number of moles in 5.75 gm of sodium. (Na=23u)
(ii)Define law of constant composition.
(iii)What is atomicity of chloroform and methane.
Question 16 (3 Marks)
(i)What are infectious diseases? Give two examples.
(ii)Name two infectious agents.
Question 17 (5 Marks)
(i)A gold sample contains 90% of gold and rest copper. How many atoms of gold are present in one gram of this sample of gold. (Au=197u)
(ii)Which has more number of atoms, 100 g of calcium or 100 g of oxygen gas. (Ca=40 u, O=16u)
Question 18 ( 5 Marks)
A mass of 100 Kg is dropped from a height of 50 cm. Find its
(i)Potential energy just before dropping
(ii)Kinetic energy on touching the ground.
Question 19 (5 Marks)
(i)State a condition for an echo to be heard.
(ii) Bats cannot see, then how do they catch their pray?
Question 20 (5 Marks)
(i)Name two diseases against which infants below one year are vaccinated.
(ii) Why are antibiotics effective against bactaria but not against viruses.
Question 21 (5 Marks)
(i)A sound wave travels at a speed of 339 m/s. If the wavelength is 1.2 cm, what is the frequency of the wave?
(ii) If 20 waves are produced per second, what is the frequency in hertz?
Question 22 (5 Marks)
(i)Distinguish between acute and chronic diseases.
(ii)What will be the symptom of a disease if the target organs are
(a)Lungs
(b)liver
Question 23 ( 5 Marks)
Drive an expression to calculate energy for an object in motion and calculate the work required to stop a car of mass 1500 Kg moving at a velocity of 60 KM/hr
OR
(i)Name and define the physical quantity whose SI unit is watt. Write the relation between kWh and joule.
(ii)A pump delivers 1000 L of water in a tank at a height of 15 m in 120 sec. Calculate work done by the pump and its power. (Take g=9.8 m/s2)
Question 24 (5 Marks)
- Describe briefly the Rutherford’s alpha particle scattering experiment. Write the important observations and conclusions drawn from the experiment.
- If the bromine atom is available in the form of two isotopes ${}_{35}^{79}Br$ and ${}_{35}^{81}Br$ in 49.7% and 50.3% abundance respectively. Calculate the average atomic mass of bromine.
OR
- Define the atomic number and atomic mass number of element.
- One atom of an element contains 8 protons and 9 neutrons. Predict
- Number of electrons
- Atomic number
- Atomic mass
- An isotope for this element
- Write the main postulates of Bohr’s atomic model.
Question 25 (5 Marks)
Describe the steps and process involved in the nitrogen cycle. Also show cycling of various nutrients in this cycle diagrammatically.
OR
- How do forests influence the following
- Air
- Soil
- Water
- How are CFC’s harmful for the environment and living beings?
Section B ( 1 Marks )
Question 26 The correct step shown for an experiment to establish relationship between loss in weight of an immersed solid with the weight of water displaced by it is:
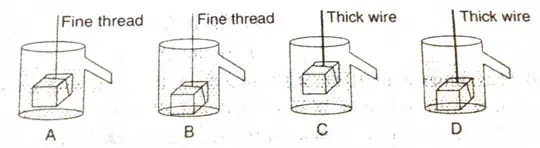
- A
- B
- C
- D
Question 27 The mass of a solid iron cube of side 4 cm is to be determined using a spring balance. If the density of iron is approximately 8.5 gcm-3 , the best-suited spring balance for determining the weight of the solid would be of:
- Range 0-250 gwt ; least count 1 gwt
- Range 0-250 gwt ; least count 5 gwt
- Range 0-1000 gwt ; least count 5 gwt
- Range 0-1000 gwt ; least count 10 gwt
Question 28 A body is weighted in liquid by immersing it fully in each of the three containers shown below. The apparent weight of the solid will be
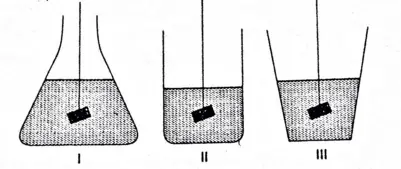
- Least in I
- Least in II
- Least in III
- Equal in all
Question 29 The correct way of reading a liquid level is shown in:
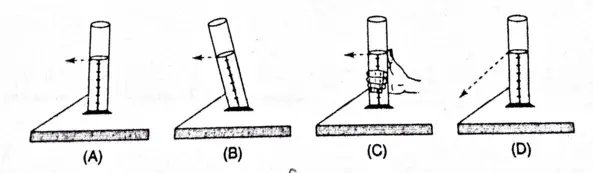
- Figure A
- Figure B
- Figure C
- Figure D
Question 30 A glass cuboidal has dimensions $10cm\times 10cm\times 4cm$ . It is kept with its face $10cm \times 10cm$ . If it is lifted and allowed to rest on the table with its smaller surface $10cm \times 4cm$ in contact with the table the pressure exerted will
- Increase
- Decrease
- Remain unchanged
- May increase or decrease depending on the shape of the table
Question 31 An iron cuboid and a wooden cuboid of the same dimensions are placed on the sand on the face having the same area, then
- Iron cuboid applies greater pressure
- Both cuboid applies same pressure
- Wooden cuboid applies greater pressure
- They exert no pressure
