Oxygen is denoted by the letter O and its atomic number is 8. The number of electrons in Oxygen is 8. It is a highly reactive non-metal. We will checking about Electron configuration of oxygen
Electron configuration of Oxygen
Electron configuration is the Arrangement of electrons in different orbitals of an atom. The electronic configuration of Oxygen is given by
1s2 , 2s2, 2p4
or
2, 6
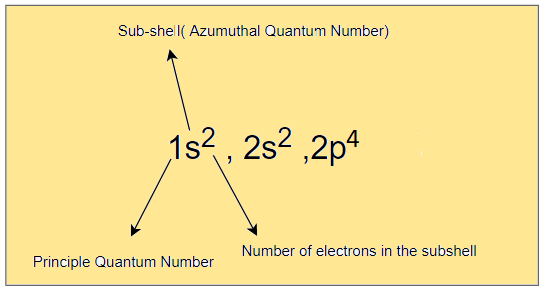
we can write the electron configuration in the orbital diagram as below
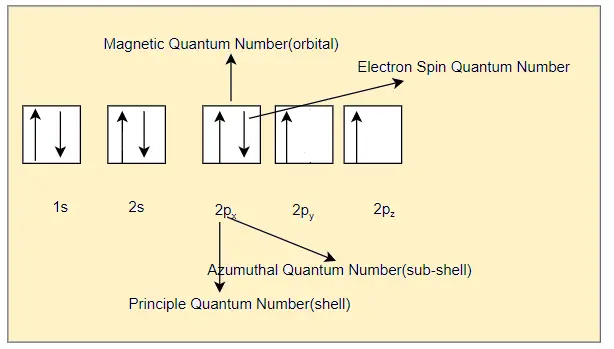
Sometimes, we can write the electron configuration in short form as
[He] 2s2 2p4
Where [He] stands for electron configuration of Helium i.e 1s2
I hope you like this article on Electron configuration of oxygen
Related Articles
Electron configuration of calcium
Electron configuration of sodium
Electronic Configuration
