James Prescott Joule (1818 – 1889)
- He was an English physicist and brewer, born in Salford, Lancashire.
- He received his early education at home
- He studied for sometime under John Dalton
- Joule studied the nature of heat, and discovered its relationship to mechanical work .
- His first major research was concerned with determining the quantity of heat produced by an electric current and, in 1840, Joule discovered a simple law connecting the current and resistance with the heat generated.
- In 1848 Joule published a paper on the kinetic theory of gases, in which he estimated the speed of gas molecules.
- From 1852 he worked with William Thomson (later Lord Kelvin) on experiments on thermodynamics. Their best known result is the Joule–Kelvin effect – the effect in which an expanding gas, under certain conditions, is cooled by the expansion.
- The SI derived unit of energy, the joule, is named after him.
- Joule discovered the first law of thermodynamics, which is a form of the law of conservation of energy
- He Died on October 11, 1889 (aged 70) at Sale, Cheshire, England
Ludwig Eduard Boltzmann (1844-1906)
- He was an Austrian physicist,born in Vienna, then capital of the Austrian Empire on February 20, 1844
- He received his primary education from a private tutor at the home of his parents-
- He obtained his doctorate from the University of Vienna and thereafter taught at several German and Austrian universities
- Boltzmann made important contributions to the kinetic theory of gases and to statistical mechanics—the Boltzmann constant, which relates the mean total energy of a molecule to its absolute temperature, is used widely in statistics and is named for him.
- He explained the second law of thermodynamics by applying the laws of mechanics and the theory of probability to the motions of atoms, and he is remembered as the developer of statistical mechanics
- In 1863 he entered the University of Vienna, where one of his teachers was Joseph Stefan, author of the law of radiation named after him
- He Died on September 5, 1906 at Duino near Trieste, Italy (at that time Austrian Empire)
Thomson, Baron Kelvin, William(1824-1907)
- William Thomson was born on June 26, 1824, at Belfast, Ireland.
- His father taught mathematics and joined the University of Glasgow faculty in 1831.
- He devoted much of his time to the education of his sons and arranged for them to audit university classes. Thus when William was 10, he matriculated in the university and did well. After a tour of the Continent, Thomson entered Peterhouse, Cambridge, in 1841.
- He did important work in the mathematical analysis of electricity and thermodynamics, and did much to unify the emerging discipline of physics in its modern form. He is widely known for developing the Kelvin scale of absolute temperature measurement.
- With James Prescott Joule, Kelvin discovered that gases cool when allowed to expand, the Joule-Thomson effect.
- The title Baron Kelvin was given in honour of his achievements, and named after the River Kelvin, which flowed past his university in Glasgow, Scotland.
- He died on 17 December 1907 at Largs, Ayrshire, Scotland, United Kingdom
Johannes Diderik Van der Waals(1837–1923)
- Johannes van der Waals was born on Nov. 23, 1837, in Leiden, the son of Jacobus van der Waals and Elizabeth van den Burg.
- He was largely self-taught in science and he originally worked as a school teacher.
- He studied from 1862 to 1865, earning degrees in mathematics and physics. He was married to Anna Magdalena Smit and had three daughters and one son.
- In 1866, he became director of a secondary school in The Hague. In 1873, he obtained a doctorate degree in Leiden under Pieter Rijke. In 1876, he was appointed the first professor of physics at the newly established University of Amsterdam.
- he extended the classical ideal-gas law to describe the behaviour of real gases, deriving the van der Waals equation of state in 1881.
- His work led to the liquefying of several common gases and made possible the study of temperatures near absolute zero.
- The weak electrostatic attractive forces between molecules and between atoms are called van der Waals forces in his honor.. He received a 1910 Nobel Prize.
- Van der Waals died in Amsterdam in 1923, one year after his daughter’s death.



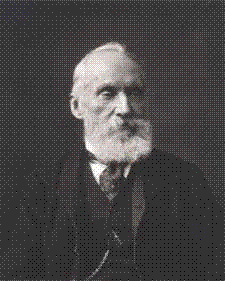

thankss for giving the use ful information 2 us
great you like it.. we always try to help people out with their problems in physics…