

We have already defined Heat Conduction while learning about heat transfer.
Heat conduction is the transfer of heat from the hotter part of the material to its colder part without the actual movement of the particles.
During the heat conduction process heat energy is transferred only to the successive molecules. In this process, molecules do not leave their mean position at all. So in conduction particles of the medium transfers heat without leaving the mean position.
Heat conduction does not take place in liquids and solids. This happens because molecules of both gases and liquids move randomly or in a specific direction.
When one end of a metal bar is heated, molecules at this end begin to vibrate faster and faster. These particles at the hot end have a greater kinetic energy of vibration in compassion to the particles that are not directly under heat exposure. These faster moving particles/molecules collide with their neighboring molecules. Here from the figure you can see how heat is being transferred at the molecular level through the vibrations of molecules at heated end to the colder end.
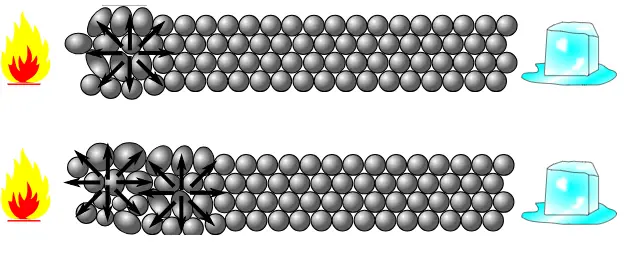
Solid substances which conduct heat easily are good conductors of heat. For example silver, copper etc. All metals are a good conductor of heat.
Those substances which do not conduct heat easily are called bad conductors of heat. For examples of wood, cloth, air, paper etc.
Generally, good conductors of heat are also good conductors of electricity.

To understand the steady state of a substance let us consider a solid metal bar. We now heat one end of the metal bar. This way one end of the rod is at a higher temperature and another end of the rod is at a lower temperature.From the discussion about the transfer of heat by conduction, we know that heat energy is transferred only to the successive molecules. Let us divide the solid metal bar into various segments as shown below in the figure
Here if the metal bar is not insulated heat is lost to the atmosphere by convection and radiation. This can be prevented by placing some non conducting material around the bar. Here if the metal bar is not insulated heat is lost to the atmosphere by convection and radiation. This can be prevented by placing some non conducting material around the bar.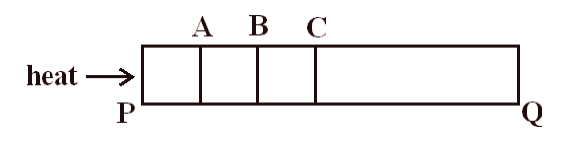
Again consider sections A, B and section C. Now section B gets heat from section A. Some part of heat received is absorbed due to which temperature of section B increases. Some part of this heat received flows to the atmosphere. Remaining part of the heat is transferred to adjacent section C.
In this state temperature of every cross-section of the bar. Goes on increasing with time. This is the transient or variable state of heat conduction.
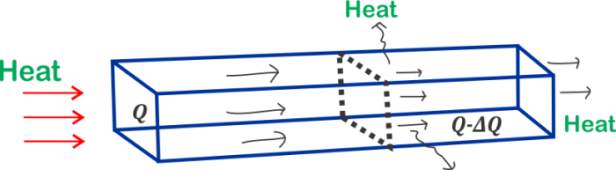
This state of the bar when the temperature of every cross-section of the bar becomes constant and there is no further absorption of heat in any part is called Steady-State or Stationery-State.It is important to note here that
The surface of a substance whose all particles or molecules are at the same temperature is called an isothermal surface.Figure 5 below shows two isothermal surface $P$ and $Q$. Here temperature of all points on surface $P$ is same. Temperature of surface $P$ is not same as the temperature of surface $Q$.
The rate of change of temperature with distance in the direction of flow of heat is called temperature gradientLet us consider the figure given below
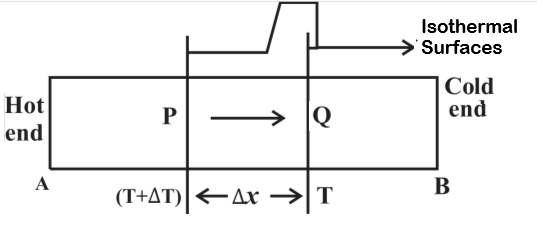
Above figure shows two isothermal surfaces P and Q. Surface P is at temperature $(T+\Delta T)$ and surface Q is at temperature $T$.
The quantity of heat transferred from an isothermal region per unit time in the direction of heat propagation is called the rate of heat flow. It is denoted by $H$. It is also known as heat current.
If $Q$ is the amount of heat transferred in time t; then
$$H=\frac{Q}{T}$$
Units are $Kcal{s ^{ - 1}} $ , $J{s ^{ - 1}}$ and $cal{s ^{ - 1}}$
The rate of heat flow per unit area is called the specific rate of heat flow. It is denoted by $q$.
$q=\frac{H}{A}$
Units are $Kcal.{m^{ - 2}}{s^{ - 1}}$, $K{m^{ - 2}}{s^{ - 1}}$ and $Cal.cm^{ - 2}{s^{ - 1}}$